Performance of Galvanized Steel in Different Environments
Does hot-dip galvanized steel perform the same in any type of environment?
Hot-dip galvanized steel is specified in many different applications and environments. Exposure conditions include atmospheric, submerged applications, chemical environments, and soil. Although the galvanized coating provides a barrier and cathodic protection in each of these environments, the mechanism of corrosion protection can be different. Lets explore how the hot-dip galvanized coating protects the underlying steel in different environments.
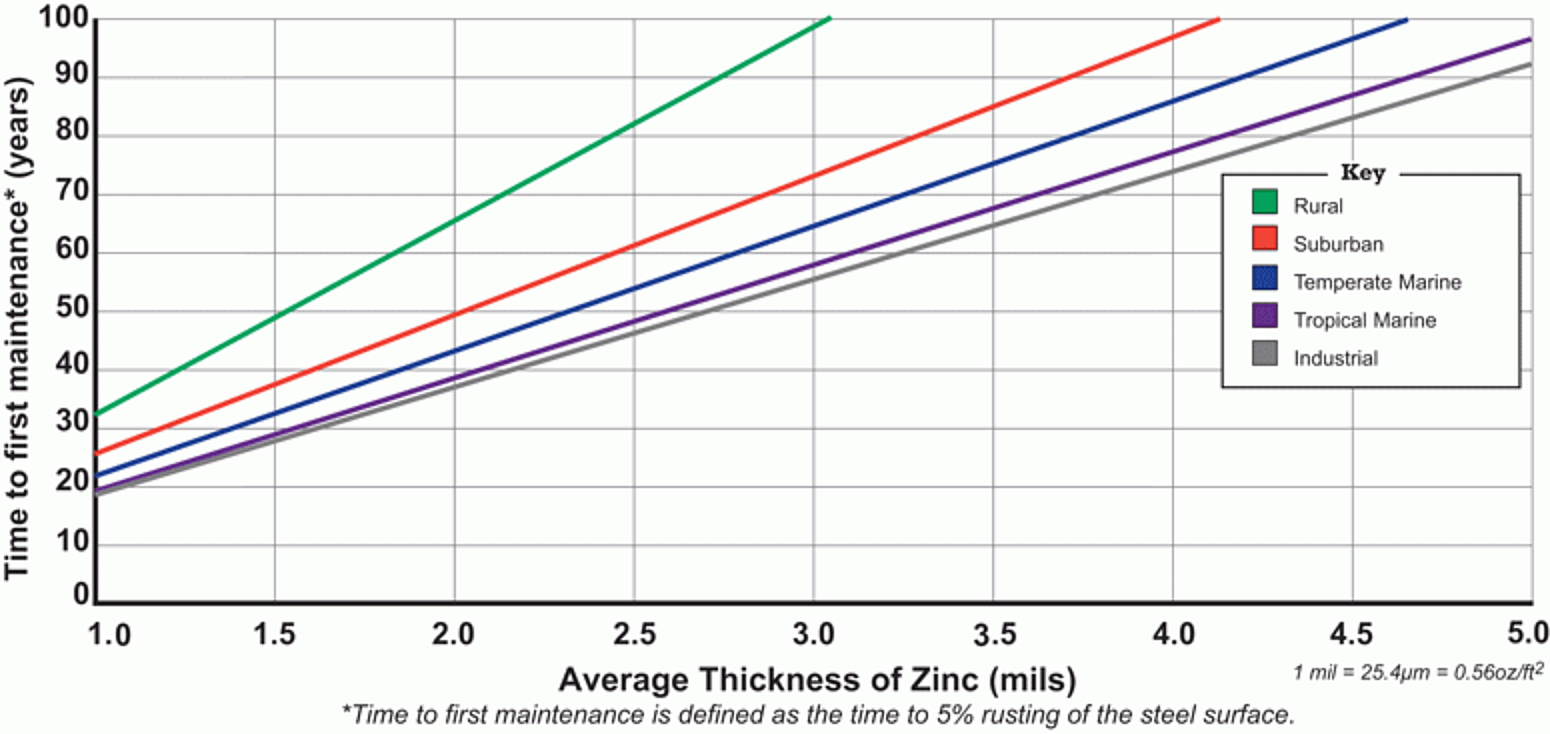
In the Atmosphere
Zinc, like all metals, naturally corrodes when exposed to the atmosphere. The corrosion products that form on the zinc surface provide a passive, impervious barrier. Three different products build on the surface over time to develop what is collectively known as the zinc patina. Zinc oxide is the initial corrosion product on the surface and is formed by a reaction between the zinc coating and oxygen in the atmosphere. When the zinc oxide interacts with moisture, it can be converted to zinc hydroxide. The zinc hydroxide and zinc oxide further react with carbon dioxide in the air to form zinc carbonate. The zinc carbonate particles form a layer of tightly adherent and relatively insoluble particles on the surface. The layer, or patina, is primarily responsible for the long-lasting corrosion protection provided by the galvanized coating in most atmospheric environments.
The corrosion behavior of the galvanized coating in various atmospheric environments is influenced by many factors such as prevailing wind direction, type and density of corrosive fumes and pollutants, amount of sea spray, number of wetting and drying cycles, and the duration of exposure to moisture. Although there is a range in observed corrosion rates, actual observed rates rarely exceed 0.3 mils per year.
The American Galvanizers Association (AGA) has compiled corrosion data from real-world test sites into the Time to First Maintenance Chart, as shown in Figure 1. The corrosion data is separated into five environmental classifications, which include rural, suburban, temperate marine, tropical marine, and industrial. These classifications are the types of atmospheric environments in which hot-dip galvanized steel is normally specified. A rough time to first maintenance estimate can be obtained from the chart based on the of the coating thickness and the environmental classification.
In Soil
Coarse and textured soils, such as gravel and sand, permit free circulation of air, and the process of corrosion may closely resemble atmospheric corrosion. Clay and silt soils have a fine texture and hold water, resulting in poor aeration and drainage. The corrosion process in such soils may resemble the corrosion process in water.
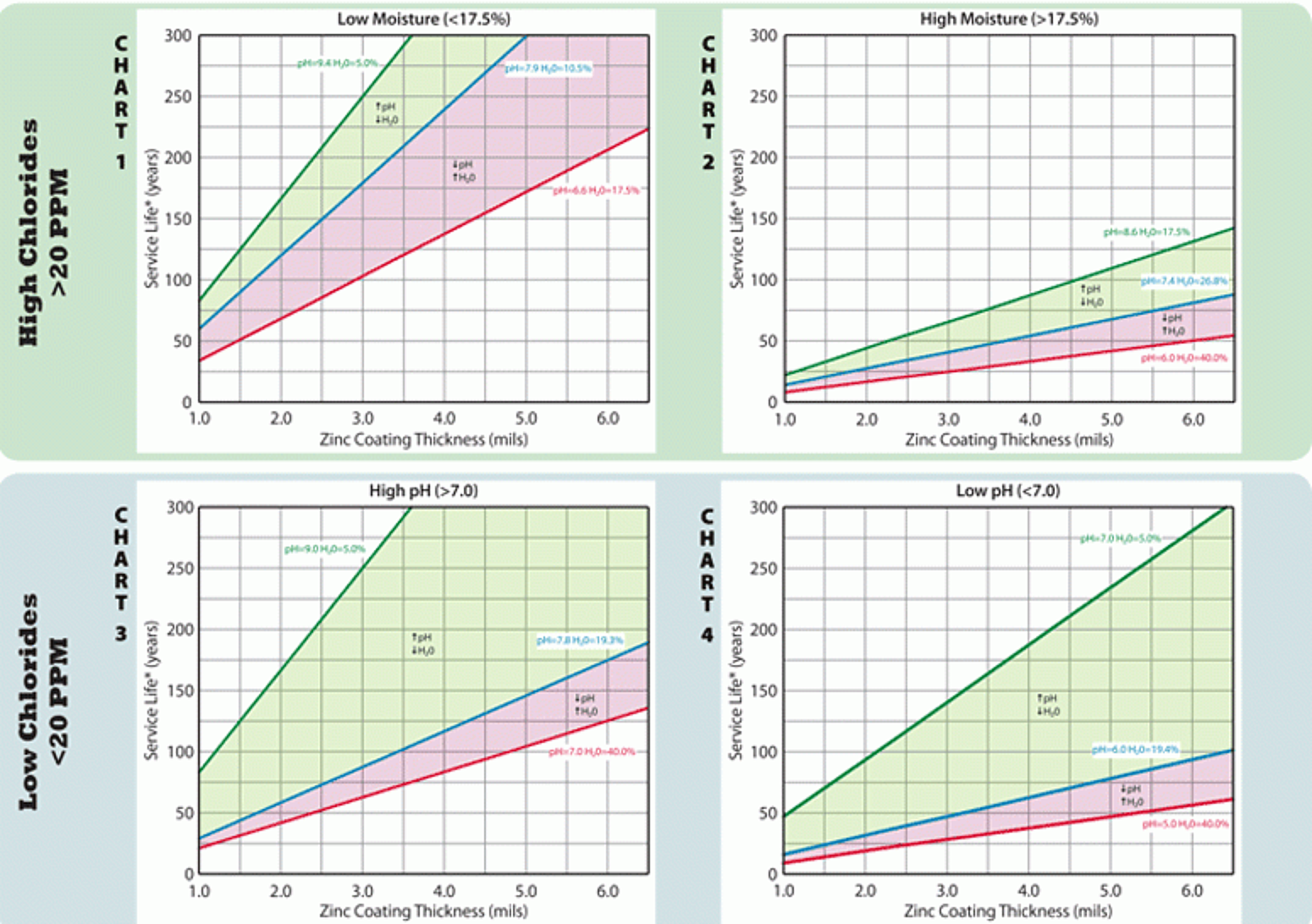
Corrosion research on different types of soil suggests a linear relationship between galvanized coating thickness and performance and the existence of four basic and distinct soil conditions, each with a different effect on buried hot-dip galvanized steel. There are critical limits for pH, water content, and chloride concentration which determine the durability of the galvanized coating. In between the limits, as pH, water content and chloride concentration vary, so too does the corrosion rate of the hot-dip galvanized steel coating. Since most galvanized structural steel has at least 3.9 to 5 mils of zinc coating, estimated service life in the harshest soil is 35 to 50 years and in the less corrosive soil, 75 years or more. The estimate of service life utilized here is years to complete corrosion of the zinc coating plus an additional time of 25%, to account for partial corrosion of the steel member. This provides the project engineer with information to determine the time to replacement or maintenance for non-critical items such as ground rods and reinforced earth wire to more critical structural components such as pilings, utility poles, and guardrail posts. The AGA has compiled corrosion data and service life estimates for hot-dip galvanized steel in soil (Figure 2).
In Water Fresh Water

Galvanizing is successfully used to protect steel in freshwater exposure. Freshwater refers to all forms of water except sea water. Freshwater may be classified according to its origin or application. Included are hot and cold domestic, industrial, river, lake and canal waters. Corrosion of zinc in fresh water is a complex process controlled largely by impurities in the water. Even rain water contains oxygen, nitrogen, carbon dioxide and other dissolved gases, in addition to dust and smoke particles. Groundwater carries micro-organisms, eroded soil, decaying vegetation, dissolved salts of calcium, magnesium, iron, and manganese, and suspended colloidal matter. All of these substances and other factors such as pH, temperature, and motion affect the structure and composition of the corrosion products formed on the exposed zinc surface. Relatively small differences in freshwater content or conditions can produce relatively substantial changes in corrosion products and rate. Thus, there is no simple rule governing the corrosion rate of zinc in fresh water. However, one trend data supports is hard water is much less corrosive than soft water. Under conditions of moderate or high water hardness, a natural scale of insoluble salts tends to form on the galvanized surface, which slows the corrosion rate of the galvanized coating.
Seawater
Galvanized coatings provide considerable protection to steel immersed in seawater. The factors that influence the corrosion of zinc in fresh water also apply to sea water. However, it is the dissolved salts (primarily sulfides and chlorides) in seawater that are the principal determinants of the corrosion behavior of zinc immersed in seawater. Given the high level of chloride in sea water, a very high rate of zinc corrosion might be expected. However, the presence of magnesium and calcium ions in seawater has a strong inhibiting effect on zinc corrosion in this type of environment.
Chemical Environments
A primary factor governing galvanized coating corrosion behavior in liquid chemical environments is the solutions pH. Galvanizing performs well in solutions of pH above 4.0 and below 12.5. This should not be considered a hard and fast rule because factors such as agitation, aeration, temperature, polarization, and the presence of inhibitors also may change the corrosion rate. Within the pH range of 4.0 to 12.5, a protective film forms on the zinc surface and the galvanized coating protects the steel by slowing corrosion to a very low rate. The protective films exact chemical composition is somewhat dependent upon the specific chemical environment.
More Information
As you can see, hot-dip galvanized steel is a versatile corrosion protection system, providing protection in many different types of environments. If you have questions about the performance of galvanized steel in a specific environment, please contact the AGA Technical Department for case studies and additional corrosion information contact the AGA Technical Department
© 2026 American Galvanizers Association. The material provided herein has been developed to provide accurate and authoritative information about after-fabrication hot-dip galvanized steel. This material provides general information only and is not intended as a substitute for competent professional examination and verification as to suitability and applicability. The information provided herein is not intended as a representation or warranty on the part of the AGA. Anyone making use of this information assumes all liability arising from such use.

